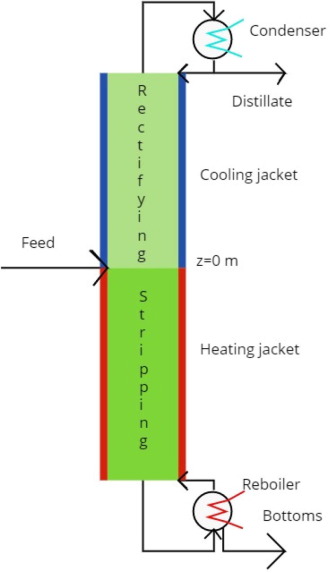
Our paper entitled Minimum entropy production in a distillation column for air separation described by a continuous non-equilibrium model has been published in Chemical Engineering Science.
In this work, we apply the rate-based model of Taylor and Krishna to describe the separation of air in a low-pressure, packed distillation column. By use of numerical optimization, we identify the temperature profile for heat exchange with the column and its surroundings that minimizes the total entropy production. Optimal operation of the column reduces the entropy production by nearly 50%, and the total heating- and cooling duties by 30% and 50%, respectively. We find that the local entropy production is more uniform for the optimal solution than in the adiabatic column, a property that may be helpful for new designs. Using the equilibrium stage model, the state of minimum entropy production has higher cooling/heating duties than in the rate-based model case. This shows that more sophisticated models can be beneficial for the development of reliable strategies to improve the energy efficiency of distillation columns.
The work was performed mainly by Diego Kingston, a visiting PhD student from the University of Buenos Aires in Argentina and in collaboration with Signe Kjelstrup from NTNU.