Our article entitled Tolman lengths and rigidity constants of multicomponent fluids: Fundamental theory and numerical examples has been accepted for publication in the Journal of Chemical Physics. The paper was very positively received by the reviewers and was chosen to be Editors pick in the journal. The work has previously drawn attention at international conferences, where my PhD student Ailo Aasen won the prize for best poster when he presented the initial results last year.
The curvature dependence of the surface tension can be described by the Tolman length (first order correction) and the rigidity constants (second order corrections) through the Helfrich expansion. In the paper, we explain the general theory for this dependence for multicomponent fluids and calculate the Tolman length and rigidity constants for a hexane-heptane mixture by use of density gradient theory. We show that the Tolman length of multicomponent fluids is independent of the choice of dividing surface and present simple formulae that capture the change in the rigidity constants for different choices of dividing surface.
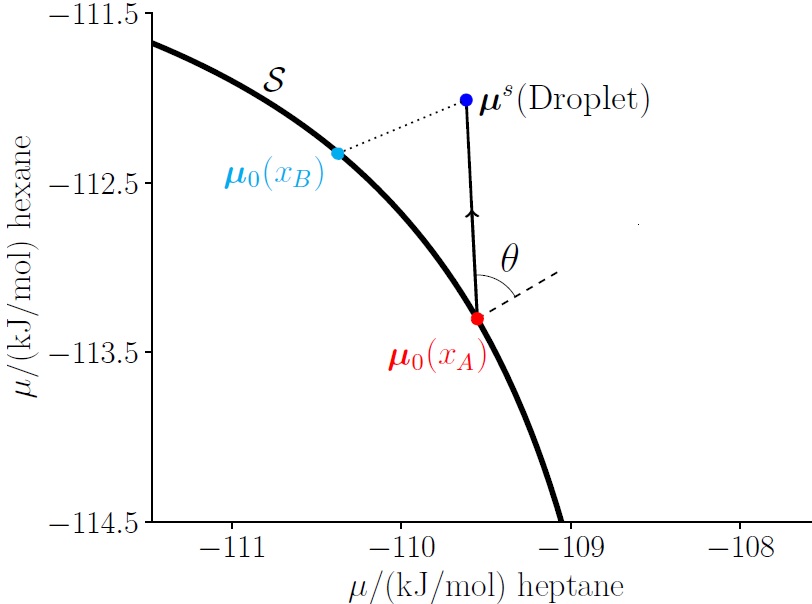
For multicomponent fluids, the Tolman length, rigidity constants and the accuracy of the Helfrich expansion depend on the choice of path in composition and pressure space along which droplets and bubbles are considered. This is illustrated in the figure below, where the chemical potential of the droplet state can be reached by both State A and State B on the saturation curve. Since the surface tension of the droplet is independent of the reference state and the surface tension of the planar surface is different for States A and B (since the surface tension depends on the composition), the Tolman length and the rigidity constants are path dependent.
For the hexane-heptane mixture, we found that the most accurate choice of path was the direction of constant liquid-phase composition. For this path, the Tolman length and rigidity constants were close to linear in the mole-fraction of the liquid-phase and the Helfrich expansion represented the surface tension of hexane–heptane droplets and bubbles within 0.1% down to radii of 3 nanometers.
The framework presented in the paper can be applied to a wide range of fluid mixtures and can be used to accurately represent the surface tension of nanoscopic bubbles and droplets, or surfaces with more exotic curvatures. A future challenge where the framework will be applied is in overcoming some of the challenges of multicomponent nucleation theory. The work was carried out in collaboration with my PhD-student Ailo Aasen and Ass. Prof. Edgar M. Blokhuis from the University of Leiden.